A Review of Periorbital Cellulitis and Infectious Conjunctivitis
RELEASE DATE
April 1, 2026
EXPIRATION DATE
April 30, 2028
FACULTY
Katherine Hale, PharmD, BCPS, MFA
Freelance Medical Writer
Tri-Cities, Washington
DISCLOSURE STATEMENTS
Dr. Hale has no actual or potential conflicts of interest in relation to this activity.
Postgraduate Healthcare Education, LLC does not view the existence of relationships as an implication of bias or that the value of the material is decreased. The content of the activity was planned to be balanced, objective, and scientifically rigorous. Occasionally, authors may express opinions that represent their own viewpoint. Conclusions drawn by participants should be derived from objective analysis of scientific data.
ACCREDITATION STATEMENT
 Pharmacy
Pharmacy
Postgraduate Healthcare Education, LLC is accredited by the Accreditation Council for Pharmacy Education as a provider of continuing pharmacy education.
UAN: 0430-0000-26-046-H01-P
Credits: 2.0 hours (0.20 ceu)
Type of Activity: Knowledge
TARGET AUDIENCE
This accredited activity is targeted to pharmacists. Estimated time to complete this activity is 120 minutes.
Exam processing and other inquiries to:CE Customer Service (800) 825-4696 or cecustomerservice@powerpak.com
DISCLAIMER
Participants have an implied responsibility to use the newly acquired information to enhance patient outcomes and their own professional development. The information presented in this activity is not meant to serve as a guideline for patient management. Any procedures, medications, or other courses of diagnosis or treatment discussed or suggested in this activity should not be used by clinicians without evaluation of their patients’ conditions and possible contraindications or dangers in use, review of any applicable manufacturer’s product information, and comparison with recommendations of other authorities.
GOAL
To review the clinical presentation and treatment of two types of ocular infections: periorbital cellulitis and infectious conjunctivitis.
OBJECTIVES
After completing this activity, the participant should be able to:
- Discuss the incidence and burden of both periorbital cellulitis and infectious conjunctivitis.
- Identify common causative pathogens of periorbital cellulitis and infectious conjunctivitis.
- Explain appropriate antimicrobial, antiviral, and supportive therapies for periorbital cellulitis and infectious conjunctivitis.
- Describe the pharmacist’s role in the prevention and management of ocular infections.
ABSTRACT: Periorbital cellulitis and infectious conjunctivitis are two types of ocular infections. Periorbital cellulitis, which affects skin and soft tissue anterior to the orbital septum, is less common than infectious conjunctivitis and is more prevalent in children than adults. Its treatment is guided by causal pathogen, predisposing factor, patient age, and infection severity. Infectious conjunctivitis, which is common and often self-limiting, is caused by bacterial or viral pathogens and accounts for one-third or more of nonemergent eye concerns. Bacterial conjunctivitis is more prevalent in children, and viral conjunctivitis is more prevalent in adults. Treatment of infectious conjunctivitis includes antimicrobial, antiviral, and supportive therapies. Aspects of pharmacist involvement in ocular-infection management include patient and provider education, medication optimization, and antimicrobial stewardship.
A reported 2% to 3% of all primary care and emergency department (ED) visits are related to eye complaints, with infection concern a common reason for seeking care.1 Although many complaints are benign and self-limiting, others may jeopardize vision or even be life-threatening. Nonemergent eye concerns accounted for 44.3% of about 12 million ED visits for ocular complaints, with more than one-third of visits related to conjunctivitis, and emergent concerns (e.g., corneal abrasion, foreign body) accounted for 41.2% of visits.2 Diagnosis and treatment of ocular infections can be challenging for providers owing to diagnostic uncertainty, concern for comorbidities and sequelae, antimicrobial-resistance patterns, and difficulties in performing the physical examination.3 Two types of ocular infections, periorbital cellulitis and infectious conjunctivitis, will be reviewed here.
ANATOMY OF THE EYE
The eye has three layers.3-5 Outermost are the cornea and sclera; the middle layer, or uvea, contains the ciliary body, iris, and choroid; and the retina is innermost. The aqueous humor, which is continuously produced by the ciliary body and reabsorbed, fills the anterior segment of the eye. The vitreous humor is a nonregenerated gel-like substance that fills the posterior eye segment. The conjunctiva, a translucent vascularized membrane, covers the visible portion of the sclera (bulbar conjunctiva) and the eyelids (tarsal conjunctiva).3-5
PERIORBITAL CELLULITIS
Periorbital cellulitis, also known as preseptal cellulitis, is an acute infection of the skin and soft tissue anterior to the orbital septum. This differs from orbital cellulitis, which occurs posterior to the orbital septum. The Chandler classification system for orbital infections and complications of sinusitis, developed in 1970 based on clinical criteria and later revised to incorporate imaging, is commonly used for assessment.6,7 Periorbital cellulitis is classified as Group I, with Groups II–V designating more severe infections. If the infection is isolated, periorbital cellulitis can potentially be managed on an outpatient basis and imaging may not be required.6,7 Owing to the severity of the infections comprising Groups II–V (orbital cellulitis with or without abscess, subperiosteal abscess, cavernous sinus thrombosis), inpatient care is required to prevent or manage severe complications.6,7
The prevalence of periorbital cellulitis in children is much higher than in adults; it occurs more frequently in those aged 2 to 6 years, with a median age of 5 years.8,9 A retrospective review of ED visits for primary or secondary diagnosis of periorbital or orbital cellulitis in the United States over a 13-year period found that the incidence rate of periorbital cellulitis increased from 6.2 per 100,000 individuals in 2006 to 19.2 per 100,000 persons in 2018, and combined ED costs for periorbital and orbital cellulitis exceeded $997 million.10 Periorbital cellulitis is more common than orbital cellulitis, as shown by two retrospective studies evaluating ED visits or hospitalizations in patients diagnosed with periorbital or orbital cellulitis.9,11 In these studies, periorbital cellulitis occurred in 80% to 86% of patients versus orbital cellulitis in 14% to 20%.9,11
Characteristics and Diagnosis
Aside from location, the infection route may also distinguish periorbital from orbital cellulitis. Orbital cellulitis is more severe than periorbital cellulitis, often arising from extended infection related to rhinosinusitis due to proximity to the paranasal sinus (particularly the ethmoid sinus) and the thinness of the orbital septum. Common periorbital infectious pathways include the hordeolum (stye), lacrimal infections (dacryocystitis/adenitis), dental abscess, periocular surgery, and breaks in the skin barrier resulting from insect bites, acne, eczema, impetigo, or other trauma.9,11,12
Patients with periorbital cellulitis will exhibit signs or symptoms of eyelid warmth, swelling, tenderness, and redness or discoloration. Fever may or may not be present. Visual acuity, ocular movement, and the globe (i.e., eyeball) are not affected. A thorough history of any surgeries (ocular, dental, sinus), sinus or dental disease, and trauma should be obtained. The presence of a nidus of skin infection should be determined, if possible. The patient should be evaluated for proptosis, vision changes, headache, facial pain, fever, pain with eye movement, restricted eye movement, photophobia, diplopia (resolving with closure of affected eye), decreased visual acuity, decreased color vision, and pupillary abnormalities.7,12-14 Assessment by an ophthalmologist or ED evaluation and/or management is recommended in the event that the provider is unable to perform an adequate physical assessment, significant soft-tissue swelling prevents assessment of the globe, or there is concern for severe infection or trauma.7
Pathophysiology
Staphylococcus aureus and Streptococcus pyogenes are the most common pathogens involved in periorbital cellulitis, with one study noting that up to 75% of cultured infections were caused by these bacteria. 8,11,15 Methicillin-resistant S aureus (MRSA) is becoming more prominent.7,8,16 Other pathogens include Streptococcus pneumoniae, Staphylococcus epidermidis, and Haemophilus influenzae type b (Hib). However, Hib vaccination has reduced the frequency of Hib-related periorbital cellulitis.8,13,17-19 Uncommon pathogens that have been linked to periorbital cellulitis include Pseudomonas aeruginosa, Proteus species (spp), Acinetobacter, Nocardia, Bacillus anthracis, Mycobacterium tuberculosis, Neisseria gonorrhoeae, and Pasteurella. Ringworm (Trichophyton)-related infection has been reported.20 Fungal infections can also cause periorbital cellulitis, although this is rare.8
Treatment
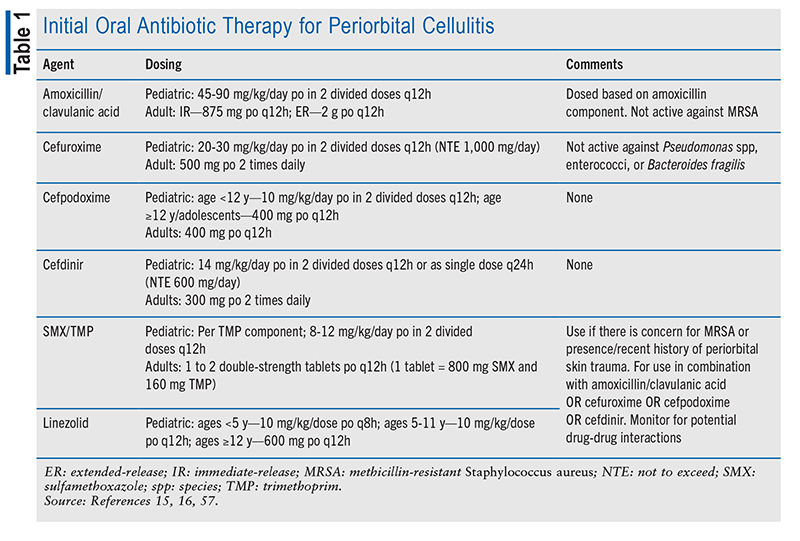
No guidelines specific to periorbital cellulitis treatment are available. Choice of therapy is guided by the causal pathogen, predisposing factors, patient age, and severity of infection. Local prevalence of MRSA should be taken into account.12 Hospitalization and broad-spectrum IV antibiotics (e.g., vancomycin with ceftriaxone or piperacillin/tazobactam) should be considered for patients with severe infection, concern for a transition to or the presence of orbital cellulitis, and age younger than 1 year (based on increased risk of worsening inflammation and/or infection owing to the eye anatomy not being fully developed).8,21 If orbital cellulitis has been ruled out, outpatient therapy may be considered for patients aged older than 1 year and with mild infection. Empiric therapy should target S aureus, Streptococcus spp, and anaerobes.8,21
Outpatient empiric therapy with amoxicillin/ clavulanic acid, cefdinir, or cefpodoxime is suggested, with the addition of sulfamethoxazole/trimethoprim (no group A streptococcus coverage) or clindamycin if MRSA is a concern.8,15 Doxycycline may be an alternative option for MRSA coverage; however, it does not cover group A streptococcus, and it should be avoided in patients aged 8 years or younger.8 A therapy duration of 5 to 7 days is recommended, but therapy may be extended if infection persists. Hospitalization and imaging should be considered if no improvement is evident within 24 to 48 hours.8,12 TABLE 1 describes oral medication options for outpatient treatment of periorbital cellulitis.
Prompt treatment is essential to prevent complications and ensure a good prognosis. However, as with many infections, the time to evaluation and treatment, concurrent health conditions, and causative pathogen may affect the prognosis. Potential complications include progression to orbital cellulitis, abscess formation, vision changes or loss due to optic nerve compression (if abscess present), spread of infection to cerebral tissue, and cavernous sinus thrombosis.8
INFECTIOUS CONJUNCTIVITIS
Infectious conjunctivitis is categorized as bacterial or viral. Overall, viral conjunctivitis is the most common form of infectious conjunctivitis, accounting for 80% of acute cases and 55% to 80% of adult cases.4,22-24 Bacterial conjunctivitis is more prevalent than viral conjunctivitis in children, whereas the opposite is true for adults.23 Studies indicate that up to 50% to 75% of pediatric conjunctivitis cases are bacterial; in adults, bacterial conjunctivitis accounts for up to 10% to 30% of cases.22,23 An analysis of 12 million U.S. ED visits over a 5-year period (2006 to 2011) found that 28% were for conjunctivitis, with more than one-half occurring in ages 0 to 5 years (54.8%), followed by 6 to 12 years (38.3%) and 13 to 18 years (27.3%).2
Extrapolations of direct and indirect costs suggest that the U.S. spends $377 million to $857 million or more annually on conjunctivitis treatment and management.25 Yearly direct and indirect cost estimates are $800 million and up to $141 million, respectively. 25-27 A single episode of infectious conjunctivitis is estimated to cost about $670.28
Characteristics and Diagnosis
No clinical features or symptoms predict viral versus common bacterial conjunctivitis.28 Many symptoms often overlap. A thorough history and examination are necessary to determine the potential cause and guide treatment.29 Onset (rapid vs. slow progression), duration, exacerbating factors, exposure risk, and characteristics of ophthalmic discharge should be evaluated. It should be determined whether presentation is unilateral or bilateral and whether initial symptoms started in one eye and then spread. The patient’s history of contact lens wear, lens type, and hygiene associated with lens use is important to know in order to assess the potential for microbial keratitis, which requires immediate medical attention. Immune status, history of other prior systemic illness that may affect the eye, immunization status, and history of recent surgery should be ascertained, and eye trauma should be ruled out.29
Physical examination of the eye and surrounding skin structures and evaluation of vision status and acuity should be conducted. Slit-lamp biomicroscopy may be performed by an ophthalmologist. An ophthalmology referral is recommended for any of the following: vision loss, moderate-to-severe pain, severe and purulent discharge, concern for corneal involvement or conjunctival scarring, episode recurrence or lack of response to treatment, history of herpes simplex virus (HSV), and/or immunocompromised status.29
Obtaining cultures of ophthalmic discharge is not recommended for mild infection but may be indicated for a recurrent, severe, or chronic infection not responsive to treatment. Adenoviral point-of-care tests, which are available but not performed routinely, have been shown to have a sensitivity of 88% to 93% and a specificity of 91% to 96%.29,30 Viral cultures, polymerase chain reaction, immunoassays, and immunochromatography have also been utilized.29,30
Identifying the clinical cause of conjunctivitis and differentiating infectious conjunctivitis from other causes of red eye are key. Goals of therapy include symptomatic relief, preventing complications, restoring or maintaining normal vision, and reducing transmission and spread.29
Pathophysiology
Causative pathogens in viral conjunctivitis include adenovirus, HSV, enterovirus, and coxsackievirus, and many other viruses have also been implicated. However, an estimated 65% to 90% of viral conjunctivitis cases are caused by the highly contagious adenovirus, which has a transmission rate of up to 50%.4,24,30 Viral conjunctivitis due to adenovirus typically occurs more frequently during summer and fall months. Multiple serotypes exist, with regional variability.4
Adenovirus spreads via contaminated objects such as the hands, medical instruments, swimming-pool water, fomites, eye solutions, makeup, and personal items.4,22,24,30 The incubation period is 10 to 14 days. The viral course is about 10 days, and viral shedding may occur for up to 2 weeks. Because of this, limiting contact with others for 10 to 14 days is recommended. Not only is adenovirus highly transmissible, it can persist for long periods of time on common surfaces, reportedly present for as long as 4 to 5 weeks.4,24,30,31 Adenoviral conjunctivitis may lead to pharyngoconjunctival fever and epidemic keratoconjunctivitis (EKC).22,29 In the event of EKC, it is recommended that the patient wait until active infection is resolved to return to work or school.30
Treatment
Adenoviral conjunctivitis treatment is primarily supportive, as no specific therapy has been shown to eradicate adenovirus infection. Symptomatic treatment with cold compresses, artificial tears and lubricating eye drops, and topical antihistamines may provide some relief.3,4,29 Topical corticosteroids may be indicated short-term in severe cases to improve symptoms, reduce scarring, and improve vision in patients with subepithelial corneal infiltrates; indiscriminate use should be avoided. Prescribing and management should be performed only by an ophthalmologist, with close monitoring needed owing to concerns over prolonged viral shedding, increased intraocular pressure, reactivation or worsening of HSV, and difficulty weaning off.24,29,30,32 Corticosteroids used in the treatment of ocular inflammation include dexamethasone, fluorometholone, loteprednol, and prednisolone preparations as topical eye drops.32 Follow-up is recommended in 2 to 3 days or 1 to 2 weeks pending examination and clinical assessment. Two- to 3-week follow-up is recommended for any patient with unresolved symptoms.29
HSV Conjunctivitis
HSV conjunctivitis is estimated to cause 1.3% to 4.8% of acute conjunctivitis cases.22,24 In the U.S., a neonatal HSV incidence rate of 15.7 per 100,000 live births has been reported.33 Skin-eye-mouth HSV disease has been demonstrated to account for up to 47% of neonatal HSV cases.34 Primary exposure to an infected individual and reactivation of a prior HSV infection owing to increased stress, other viral illnesses, surgery, trauma, or ultraviolet light exposure increase the risk of HSV conjunctivitis.29 HSV conjunctivitis may lead to epithelial or stromal keratitis, corneal edema, endotheliitis, scarring, perforation, uveitis, or retinitis.29 Symptom presentation is typically unilateral, with watery discharge, a mild conjunctival follicular reaction, and swelling of the periauricular lymph node. A vesicular rash or eyelid ulceration is considered a distinctive sign. Unless complications develop, HSV conjunctivitis is self-limited, lasting 4 to 7 days without treatment.29
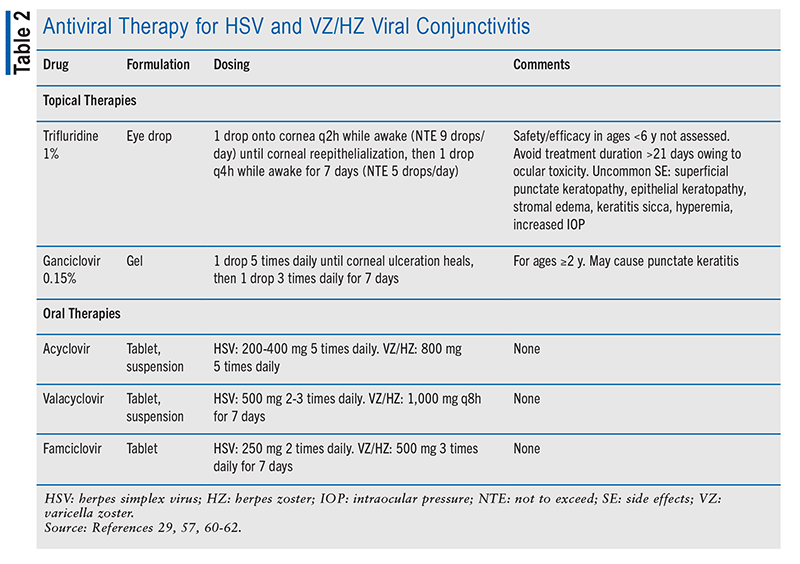
To prevent complications, HSV conjunctivitis may be treated with topical or oral antiviral therapies (TABLE 2).29 Topical options include trifluridine 1% solution, acyclovir 3% ointment, and ganciclovir 0.15% gel. Oral acyclovir, valacyclovir, or famciclovir may be utilized both for acute treatment and as long-term prophylaxis (in lower doses). The dosing of these agents must be appropriate to the patient’s renal function.15 The use of topical corticosteroids should be avoided owing to the risk of HSV reactivation or potentiation. Follow-up of 1 week is recommended.22,29
Varicella Zoster and Herpes Zoster Conjunctivitis
Varicella zoster (VZ; primary infection) virus and herpes zoster (HZ; reactivation of VZ) virus are other known causes of viral conjunctivitis. When HZ involves the eye, infection of the conjunctiva and eyelids is most common (41.1% and 45.8%, respectively), and complications may occur in 35% or more of patients.22,24,29,35 Vaccination with varicella and zoster vaccines has reduced viral incidence and sequelae, and it is recommended. Infection length is limited, lasting only a few days regardless of primary VZ or recurrent HZ infection.29
Presentation is frequently unilateral and similar to HSV. Punctate keratitis (primary VZ) or pseudodendritic keratitis (recurrent HZ) may also be present. Distinctive signs of VZ or HZ conjunctivitis include eyelid ulceration with pain, a vesicular dermatomal rash, and pleomorphic or nonexcavated pseudodendritic epithelial corneal keratitis.29
Oral antiviral therapy with acyclovir, valacyclovir, or famciclovir is the primary treatment for VZ or HZ conjunctivitis; topical antivirals are not effective, and the risk of corneal or uveal involvement is high.29 Their dosing must be appropriate to the patient’s renal function.15 Topical antibiotics may be indicated to prevent bacterial infection that can lead to severe conjunctival scarring. Long-term prophylaxis may be indicated for HZ if chronic sequelae occur.4,22,29
Bacterial Conjunctivitis
Types of bacterial conjunctivitis include nongonococcal (most common), gonococcal, and chlamydial. Bacterial conjunctivitis is also categorized as acute, hyperacute, or chronic. In acute nongonococcal bacterial conjunctivitis, S aureus is the most common pathogen in adults, whereas Hib is most common in children. Other causative pathogens include Streptococcus and Moraxella catarrhalis. 4,22,36-38 Increasing rates of MRSA conjunctivitis have been noted.22,36 Bacterial conjunctivitis may develop secondary to bacterial otitis media, pharyngitis, or sinusitis, nasolacrimal duct obstruction, severe tear deficiency, immunosuppression, or trauma.30 Transmission occurs via contaminated hands, fomites, nasopharynx, and exposure to an infected individual.4,22,29,36
Patients with nongonococcal bacterial conjunctivitis may present with symptoms occurring in one or both eyes, as infection can spread from one eye to the other, usually within 1 to 2 days. Watery discharge and redness (conjunctival hyperemia) are often the first symptoms, with progression to purulent or mucopurulent discharge within 1 to 2 days.3,4,23,29,36 Collection of debris at the eyelashes results in eyelid mattering and adherence. Itching does not typically occur. An incubation period of 1 to 7 days and a communicability period of 2 to 7 days have been estimated.22,38 The natural infection course is often mild and self-limiting, with resolution of symptoms within 7 days if left untreated.4,29,36
Because of the self-limiting nature of mild bacterial conjunctivitis, spontaneous resolution without treatment can occur. Therapy initiation should be guided by clinical assessment, expected course of infection if untreated, and antibiotic resistance. Symptom improvement, a reduced infection duration of 1 to 2 days, and a decrease in communicability have been shown with broad-spectrum topical antibiotic therapy.29 In patients aged 1 month and older, topical antibiotics were found to improve resolution of clinical signs or symptoms by 26% versus placebo (risk ratio [RR] 1.26; 95% CI, 1.09-1.46; n = 1,474), regardless of treatment duration or antibiotic class.37 The microbiological cure rate likely improved upon completion of therapy (RR 1.53; 95% CI, 1.34-1.74; n = 2,827) compared with placebo. The likelihood of persistent clinical infection also decreased. Eligibility criteria, outcomes assessed, assessment timepoints, and intervention (antibiotic used, dose, duration) led to trial heterogeneity, limiting study results.37
Exclusion from school/childcare is not recommended for mild bacterial conjunctivitis unless systemic symptoms are present or the infection is moderate to severe.29 In general, exclusion policies for children are variable and not clear for adults. In the U.S., 10 states have no exclusion policy for school/ childcare, five allow return 24 hours after starting antibiotic therapy, and five require provider approval. The remaining states either have unclear policies or refer to the CDC or American Academy of Pediatrics.39-41
Management approaches include prescribing of topical antibiotic therapy or the “watch and wait” approach, prescribing topical antibiotics if symptoms do not improve after a period of time (e.g., 3 days). Determining bacterial versus viral conjunctivitis can be challenging, leading to potential antibiotic overprescribing and resistance. Indiscriminate use of topical antibiotics is not recommended.29 The cost-effectiveness of antibiotic therapy and return-to-school/ childcare strategies are not fully elucidated. One U.S. study determined that a combined approach of allowing children with no systemic symptoms to return to school and refraining from topical antibiotic prescribing produced an estimated 1.6 million averted antibiotic courses and $783 million in annual savings.40 Usual care remained the most expensive ($212.73/ episode), and the combined approach was least costly ($127.38/episode).40
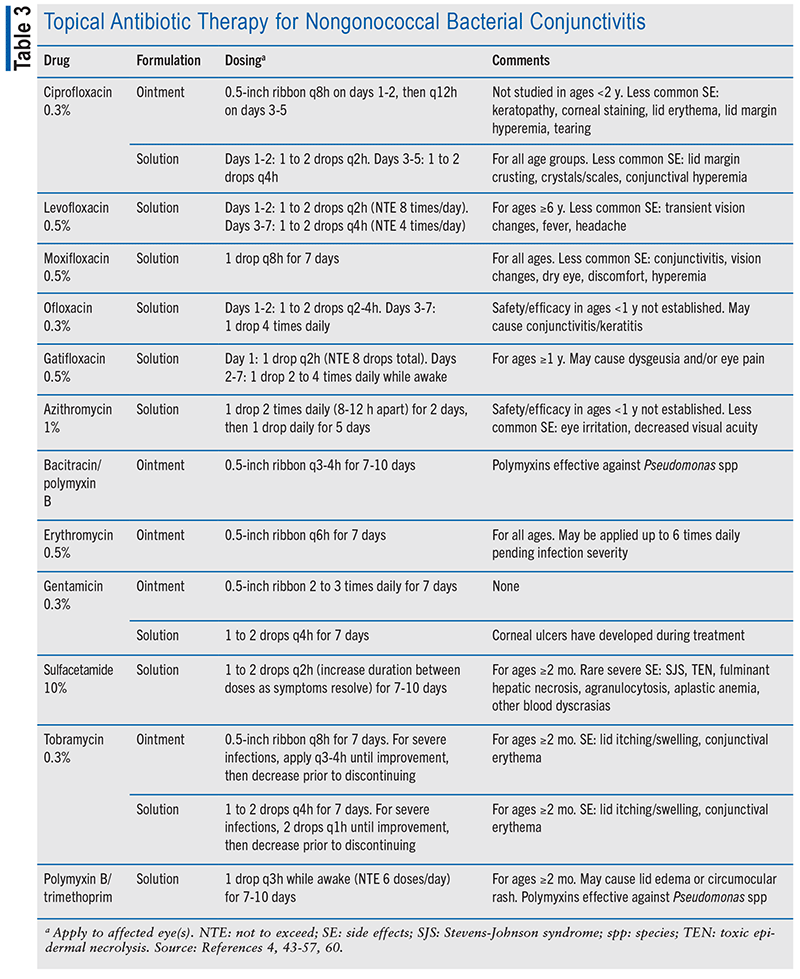
Empiric treatment for 5 to 7 days with a broad-spectrum topical antibiotic is recommended for mild infection, and a variety of broad-spectrum fluoroquinolone and nonfluoroquinolone antibiotic ointments and drops are available (TABLE 3). No one antibiotic therapy is preferred over another. In moderate-to-severe infection, conjunctival cultures may be obtained to guide therapy and rule out a gonococcal cause.29 Follow- up is recommended within 3 to 4 days if symptoms do not resolve. Common side effects of topical therapy include mild eye irritation (stinging, burning, itching), blurred vision, tearing, and foreign-body sensation. Some therapies may cause worsening of conjunctivitis or conjunctival hyperemia.42-56
MRSA resistance in ocular isolates and macrolide resistance (azithromycin) in S aureus and S pneumoniae have been observed.29,56 Fluoroquinolone therapies are most effective against gram-negative bacteria and are effective against group B streptococus. Fluoroquinolones have intermediate activity against staphylococcal infections, do not have activity against group A streptococcus, and are effective against Hib.47
Gonococcal Conjunctivitis
Gonococcal conjunctivitis due to N gonorrhoeae transmits via oculogenital spread. It typically presents in one or both eyes as hyperacute bacterial conjunctivitis in adults (owing to very rapid onset of reduced vision) and severe and copious purulent discharge. The risk of corneal involvement and potential corneal infection, perforation, and scarring is high. In neonates, symptoms present within 2 to 7 days post birth and then rapidly evolve into severe purulent infection. If preventative topical antibiotics were utilized, symptom development may be delayed.4,5,22,29,58 Gonococcal conjunctivitis in adults is uncommon, and referral to an infectious-disease specialist is recommended.59 Ophthalmia neonatorum is a severe form of gonococcal infection that can affect neonates and result in blindness. Ocular prophylaxis and routine screening programs have been implemented to prevent ophthalmia neonatorum.58
Ceftriaxone 1 g administered IM is the recommended treatment for gonococcal conjunctivitis in adults and adolescents.58 For neonates, preventative therapy with a one-time application of erythromycin 0.5% ophthalmic ointment at birth is recommended. If ophthalmia neonatorum develops, ceftriaxone may be administered IV or IM as a single dose of 25 to 50 mg/kg/body weight (maximum dose, 250 mg).29,58 Currently, the CDC recommends cotreatment for chlamydia if gonococcal infection is present.58 Daily monitoring is recommended until resolution of symptoms occurs.29
Chlamydial Conjunctivitis
Chlamydial conjunctivitis can affect both adults and neonates. Its transmission occurs via oculogenital spread, direct or indirect contact with secretions, the hands, and fomites. Chlamydia trachomatis serotypes D–K are sexually transmitted, and recurrent infection and chronic inflammation can progress to chlamydial inclusion. Coinfection with gonorrhea should be considered. 29,58 Neonates present with symptoms 5 to 19 days after birth, with infection occurring through vaginal delivery and perinatal cervical exposure to the infected mother. If left untreated, infection can last 3 to 12 months.29,58 As with gonococcal conjunctivitis, neonatal chlamydial conjunctivitis may progress to ophthalmia neonatorum; therefore, routine screening programs have been implemented.58
In adults, one or both eyes may be affected, presenting with follicles on the bulbar conjunctiva and semilunar fold, chemosis, papillary hypertrophy, mucopurulent discharge, tender periauricular lymph node(s), corneal pannus, and superficial keratitis. Neonates may exhibit purulent, mucopurulent, or blood-stained discharge and no follicles.3,29
Systemic therapy is the recommended treatment for chlamydial conjunctivitis, owing to likelihood of chlamydial infection also being present in other body areas.58 A 7-day regimen of doxycycline 100 mg twice daily is recommended for adults and adolescents. Alternatively, oral azithromycin given as a 1-g single dose or a 7-day course of oral levofloxacin 500 mg may be used. For neonates and infants weighing less than 45 kg, a 14-day course of oral erythromycin base or ethyl succinate administered at a dosage of 50 mg/ kg body weight/day in four divided doses is recommended. Alternatively, a 3-day course of oral azithromycin suspension at a dosage of 20 mg/kg/body weight/day once daily may be used. Topical erythromycin ointment for prevention of ophthalmia neonatorum is no longer effective against chlamydia; therefore, robust screening and prevention programs are recommended for pregnant women.29,58,59 Cotreatment for gonorrhea is also recommended owing to concerns for concurrent infection.58
THE PHARMACIST’S ROLE
Eye complaints and infection concerns are a primary reason for seeking primary or urgent/emergent care. Infectious conjunctivitis impacts school/childcare and work, resulting in significant direct and indirect costs of diagnosis and management. Periorbital cellulitis, although less common, can progress to emergent infection, necessitating inpatient treatment.
Pharmacists play an important role in the management of ocular infections. In addition to offering patient and provider education on guideline-based therapies, they can assist with medication selection and optimization to reduce potential side effects and treatment burden and to improve adherence. Pharmacists can also monitor for treatment efficacy, side effects, and renal or hepatic function. Antimicrobial stewardship is important to reduce antibiotic overprescribing and resistance, so pharmacists can work with stakeholders and/or facilitate development and management of antimicrobial-stewardship programs in all settings.
Education on proper administration technique for ophthalmic drops and ointments is key to ensure effective therapy and prevent recurrence and progression. Pharmacists can facilitate OTC treatment selection and educate patients on supportive therapies. They can assess patients for contact lens wear and provide education on cleaning-solution use and the importance of appropriate contact lens wear. Pharmacists can also work with stakeholders or spearhead infection-control and education programs on appropriate hygienic measures to prevent the spread and transmission of infectious conjunctivitis, especially in the event of adenoviral infection.
REFERENCES
- Pflipsen M, Massaquoi M, Wolf S. Evaluation of the painful eye. Am Fam Physician. 2016;93(12):991-998.
- Channa R, Zafar SN, Canner JK, et al. Epidemiology of eye-related emergency department visits. JAMA Ophthalmol. 2016;134(3):312-319.
- Barshak MB, Durand ML, Gupta A, et al. State-ofthe- art review: ocular infections. Clin Infect Dis. 2024;79(5):e48-e64.
- Winters S, Frazier W, Winters J. Conjunctivitis: diagnosis and management. Am Fam Physician. 2024;110(2):134- 144.
- Durand ML, Barshak MB, Sobrin L. Eye infections. N Engl J Med. 2023;389(25):2363-2375.
- Chandler JR, Langenbrunner DJ, Stevens ER. The pathogenesis of orbital complications in acute sinusitis. Laryngoscope. 1970;80(9):1414-1428.
- Guerin JB, Brodsky MC, Silvera VM. Infectious and inflammatory processes of the orbits in children. Neuroimaging Clin N Am. 2023;33(4):685-697.
- Zeppieri M, Bourget D. Periorbital cellulitis. In: Stat- Pearls [Internet]. Treasure Island, FL: StatPearls Publishing; 2025 Jan-.
- Santos JC, Pinto S, Ferreira S, et al. Pediatric preseptal and orbital cellulitis: a 10-year experience. Int J Pediatr Otorhinolaryngol. 2019;120:82-88.
- Dharssi S, Taneja K, Rajaii F. Epidemiology of orbital and preseptal cellulitis in the United States: a 13-year analysis. Ophthalmic Epidemiol. 2025;32(5):553-560.
- Botting AM, McIntosh D, Mahadevan M. Paediatric pre- and post-septal peri-orbital infections are different diseases. A retrospective review of 262 cases. Int J Pediatr Otorhinolaryngol. 2008;72(3):377-383.
- Hauser A, Fogarasi S. Periorbital and orbital cellulitis. Pediatr Rev. 2010;31(6):242-249.
- Allen RC. Preseptal and orbital cellulitis. Merck Manual Professional Version. www.merckmanuals.com/ professional/eye-disorders/orbital-diseases/preseptal-and-orbital- cellulitis. Accessed March 2, 2026.
- Pelletier J, Koyfman A, Long B. High risk and low prevalence diseases: orbital cellulitis. Am J Emerg Med. 2023;68:1-9.
- Deitz E, Mangus C. Microbiology and infectious disease. In: Engorn B, Fierlage J, eds. The Harriet Lane Handbook: a Manual for Pediatric House Officers. 20th ed. Philadelphia, PA: Saunders; 2015.
- Gappy C, Archer SM. Preseptal cellulitis. In: Connor RF, ed. UpToDate. Waltham, MA: UpToDate Inc. www. uptodate.com. Accessed March 13, 2026.
- Dewey J, Kais A, Nguyen J, et al. Differences between adult and pediatric periorbital cellulitis secondary to acute rhinosinusitis. Ann Otol Rhinol Laryngol. 2025;134(11):837- 841.
- Rimon A, Hoffer V, Prais D, et al. Periorbital cellulitis in the era of Haemophilus influenzae type B vaccine: predisposing factors and etiologic agents in hospitalized children. J Pediatr Ophthalmol Strabismus. 2008;45(5):300- 304.
- Ambati BK, Ambati J, Azar N, et al. Periorbital and orbital cellulitis before and after the advent of Haemophilus influenzae type B vaccination. Ophthalmology. 2000;107(8):1450-1453.
- Velazquez AJ, Goldstein MH, Driebe WT. Preseptal cellulitis caused by trichophyton (ringworm). Cornea. 2002;21(3):312-314.
- McPherson Z, Thosar D, Donnelly A, et al. Evaluation of moderate periorbital cellulitis and home-based therapy in children (EPOCH study, Part 2): a prospective single centre cohort study. Clin Exp Ophthalmol. 2025;53(2):184- 193.
- Azari AA, Barney NP. Conjunctivitis: a systematic review of diagnosis and treatment. JAMA. 2013;310(16):1721-1729.
- Johnson D, Liu D, Simel D. Does this patient with acute infectious conjunctivitis have a bacterial infection?: the Rational Clinical Examination systematic review. JAMA. 2022;327(22):2231-2237.
- Muto T, Imaizumi S, Kamoi K. Viral conjunctivitis. Viruses. 2023;15(3):676.
- Smith AF, Waycaster C. Estimate of the direct and indirect annual cost of bacterial conjunctivitis in the United States. BMC Ophthalmol. 2009;9:13.
- Schneider JE, Scheibling CM, Segall D, et al. Epidemiology and economic burden of conjunctivitis: a managed care perspective. J Manag Care Med. 2014;17(1):78-83.
- Pepose JS, Sarda SP, Cheng WY, et al. Direct and indirect costs of infectious conjunctivitis in a commercially insured population in the United States. Clin Ophthalmol. 2020;14:377-387.
- Frost HM, Jenkins TC, Meece JC, et al. Etiology and outcomes of acute infectious conjunctivitis in children. J Pediatr. 2025;276:114368.
- Cheung AY, Choi DS, Ahmad S, et al; American Academy of Ophthalmology Preferred Practice Pattern Cornea/External Disease Panel. Conjunctivitis preferred practice pattern. Ophthalmology. 2024;131(4):134-204.
- Garcia-Zalisnak D, Rapuano C, Sheppard JD, Davis AR. Adenovirus ocular infections: prevalence, pathology, pitfalls, and practical pointers. Eye Contact Lens. 2018;44(Suppl 1):s1-s7.
- Omatsu Y, Miyazaki D, Shimizu Y, et al. Efficacy of compartmentalization in controlling an adenovirus type 54 keratoconjunctivitis outbreak on Oki Island, Japan. Jpn J Ophthalmol. 2021;65(3):423-431.
- Holland EJ, Fingeret M, Mah FS. Use of topical steroids in conjunctivitis: a review of the evidence. Cornea. 2019;38(8):1062-1067.
- Pooser M, Yuan Y, Karki S, et al. Estimated incidence of neonatal herpes simplex virus infections in the United States, 2019. Obstet Gynecol. 2024;143(5S):48s.
- Melvin AJ, Mohan KM, Vora SB, et al. Neonatal herpes simplex virus infection: epidemiology and outcomes in the modern era. J Pediatric Infect Dis Soc. 2022;11(3):94- 101.
- Yawn BP, Wollan PC, St Sauver JL, Butterfield LC. Herpes zoster eye complications: rates and trends. Mayo Clin Proc. 2013;88(6):562-570.
- Pippen MM, Le JK. Bacterial conjunctivitis. In: Stat- Pearls [Internet]. Treasure Island, FL: StatPearls Publishing; 2026 Jan-.
- Chen YY, Liu SH, Nurmatov U, et al. Antibiotics versus placebo for acute bacterial conjunctivitis. Cochrane Database Syst Rev. 2023;(3):CD001211.
- Høvding G. Acute bacterial conjunctivitis. Acta Ophthalmol. 2008;86:5-17.
- Lee T, Kuo IC. Survey of state conjunctivitis policies for school-age students. J AAPOS. 2022;26(3):115.e1-115.e5.
- Frost HM, Stein AB, Keith A, Jenkins TC. Cost-effectiveness of pediatric conjunctivitis management and return to childcare and school strategies: a comparative study. J Pediatric Infect Dis Soc. 2024;13(7):341-348.
- Oke I, Geanacopoulos AT, Subramanian SV, et al. Statelevel policies and antibiotic use for pediatric conjunctivitis. JAMA Pediatr. 2025;179(6):679-681.
- Ciprofloxacin ophthalmic solution product information. Aquebogue, NY: Altaire Pharmaceuticals Inc; 2022.
- Ciloxan (ciprofloxacin ophthalmic ointment) product information. East Hanover, NJ: Novartis Pharmaceuticals Corp; 2019.
- Erythromycin ophthalmic ointment product information. Tampa, FL: Bausch & Lomb Inc; 2025.
- Levofloxacin ophthalmic solution product information. East Windsor, NJ: Advagen Pharma Ltd; 2025.
- Moxifloxacin ophthalmic solution product information. Princeton, NJ: Fosun Pharma USA Inc; 2023.
- Ofloxacin ophthalmic solution product information. East Brunswick, NJ: Rising Pharma Holding, Inc; 2025.
- AzaSite (azithromycin ophthalmic solution) product information. Waltham, MA: Thea Pharma Inc; March 2024.
- Bacitracin zinc and polymyxin B sulfate ophthalmic ointment product information. Tampa, FL: Bausch & Lomb Inc; 2023.
- Gentak (gentamicin sulfate ophthalmic ointment) product information. Lake Forest, IL: Akorn, Inc; 2022.
- Gentamicin sulfate ophthalmic solution product information. Irvine, CA: Allergan, Inc; 2013.
- Tobrex (tobramycin ophthalmic ointment) product information. East Hanover, NJ: Novartis Pharmaceuticals Corp; 2021.
- Tobrex (tobramycin ophthalmic solution) product information. East Hanover, NJ: Novartis Pharmaceuticals Corp; 2021.
- Sulfacetamide ophthalmic solution product information. Tampa, FL: Bausch & Lomb Inc; 2023.
- Polymyxin B sulfate/trimethoprim ophthalmic solution product information. Tampa, FL: Bausch & Lomb Inc; 2023.
- Asbell PA, Sanfilippo CM, Sahm DF, DeCory HH. Trends in antibiotic resistance among ocular microorganisms in the United States from 2009 to 2018. JAMA Ophthalmol. 2020;138(5):439-450.
- Tabbara KF. Antimicrobial agents in ophthalmology. In: Tabbara K, Abu El-Asrar AM, Khairallah M, eds. Ocular Infections. Berlin: Springer; 2014:19-35.
- Workowski KA, Bachmann LH, Chan PA, et al. Sexually transmitted infections treatment guidelines, 2021. MMWR Recomm Rep. 2021;70(No. RR-4):1-187.
- Hammerschlag MR, Cummings C, Roblin PM, et al. Efficacy of neonatal ocular prophylaxis for the prevention of chlamydial and gonococcal conjunctivitis. N Engl J Med. 1989;320(12):769-772.
- Zymaxid (gatifloxacin ophthalmic solution) product information. Irvine, CA: Allergan, Inc; 2017.
- Viroptic (trifluridine ophthalmic solution) product information. New York, NY: Pfizer Inc; 2025.
- Zirgan (ganciclovir ophthalmic gel) product information. Tampa, FL: Bausch & Lomb Inc; 2025.